Vertical Turbine Technology Explained

The use of turbine technology for spirometers was first described in 19822. Since that time turbine technology has become widely used by many manufacturers and has gained a reputation for stability of calibration, low cost and ease of use. However, until now, turbine spirometers could not reach the low flow sensitivity required by the ATS/ERS guidelines3 of 0.025L/s. The SpiroConnect® turbine uses patented technology to solve this problem and now responds to beyond the requirement of 1.5L/min (0.025 L/s).
Low flow sensitivity is particularly important for the diagnosis and monitoring of COPD, a condition characterised by an extended period of low flow in the spirometry manoeuvre. The SpiroConnect® is the first turbine spirometer to overcome this limitation by a radical redesign of the turbine geometry combined with reorientation the turbine axis to lie vertically. All good quality spirometry turbines use conical V jewelled bearings to minimise friction. However the horizontal orientation of the turbine in all other spirometers is not optimal for these bearings as illustrated below:

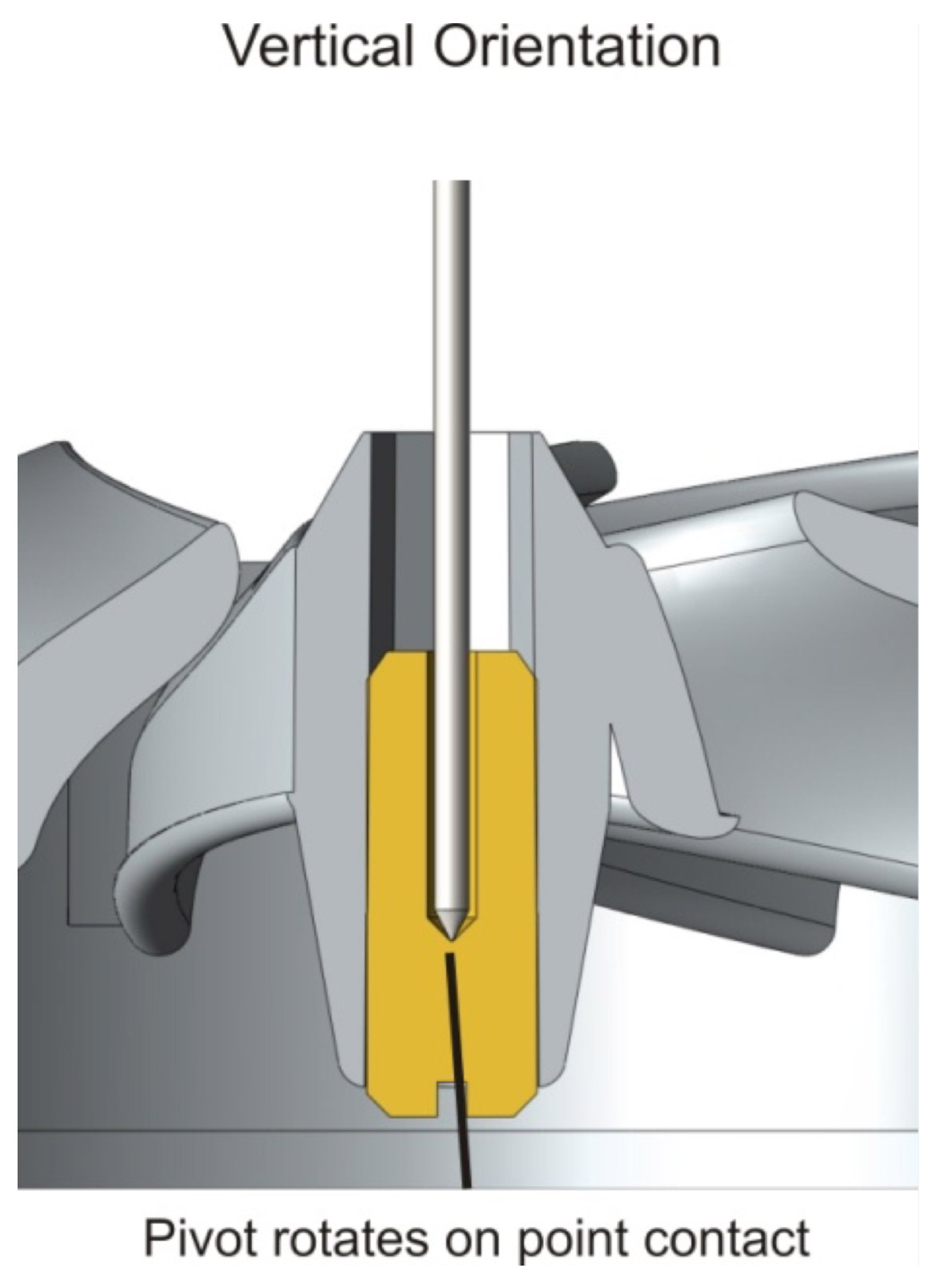
The SpiroConnect® vertical turbine obtains the optimal performance from the jewelled bearing, reducing friction and improving low flow sensitivity.
Reducing Anti Static Attraction
The effect of the static attraction between the vane and the segments of the swirl plate are reduced by curving the segments so that the attraction is distributed evenly over the whole rotational cycle of the vane.
The use of a unique anti-static plastic for the turbine eliminates any residual attraction. This attraction arises from turbo static charging which creates a small attractive force between the vane and the swirl plate. This force is negligible at high flows but significant at very low flows, impeding rotation of the vane.The plastic required for this application must be transparent to infra-red frequencies and have low resistivity. MedChip Solutions have sourced a unique plastic that is transparent to infra-red with a volume resistivity of <0.01*10-24 Ohms/m3 which is more than sufficient for the requirement.
References
1. A Comparison of devices for the Measurement of Spirometry in Normal Healthy Subjects and Patients with Respiratory Disease.
T Sharley, JA Stockley, BG Cooper Lung Function & Sleep Department, Queen Elizabeth Hospital Birmingham, B15 2GW.
2. Br Med J (Clin Res Ed).1982 Jul 3;285(6334):15-7.
Pocket-sized device for measuring forced expiratory volume in one second and forced vital capacity. Chowienczyk PJ, Lawson CP.
3. Standardization of Spirometry 2019 Update. An Official American Thoracic Society and European Respiratory Society Technical Statement.
Brian L. Graham, Irene Steenbruggen, Martin R. Miller, Igor Z. Barjaktarevic, Brendan G. Cooper, Graham L. Hall, Teal S. Hallstrand, David A. Kaminsky, Kevin McCarthy, Meredith C. McCormack, Cristine E. Oropez, Margaret Rosenfeld, Sanja Stanojevic, Maureen P. Swanney †, and Bruce R. Thompson; on behalf of the American Thoracic Society and the European Respiratory Society