Product Information
SpiroConnect®
Precision Spirometry for Diagnoses and Management of Asthma and COPD
SpiroConnect® spirometer is easy to use, extremely stable, reliable and accurate at all flows. It connects conveniently via Bluetooth to the PC, lap top computer or a Microsoft Surface tablet type device.
Its performance has been independently tested in a study aiming to establish the accuracy of spirometry measurements produced by SpiroConnect® in comparison to those obtained from a pitot-type spirometer and no clinically significant differences in the FEV1 or VC measurements occurred in the two devices.1
SpiroConnect®with its vertical turbine sensor and Bluetooth connectivity combines all known advantages of existing turbine spirometers with technological innovations to deliver precision spirometry for diagnoses and management of Asthma and COPD.
SpiroConnect® due to its patented vertical turbine design (patent number: GB2500893) is able to achieve the low flow sensitivity of 0.025 l/s required by the ATS/ERS Guidelines. This is particularly important for the diagnosis and monitoring of COPD, a condition characterised by an extended period of low flow in the spirometry manoeuvre.
SpiroConnect Data Manager® is a user friendly and intuitive PC software with functional icons and an online operating manual. It allows for a real time graphical display providing immediate patient and operator feedback and quality control, data storage, test and calibration-check report printout. It supports the following predicted value data sets: GLI GLOBAL, ECCS, Polgar, Zapletal and Austrian-Forche. The acceptance criteria can be selected from: ARTP, ATS/ERS or BTS. In the UK, SpiroConnect Data Manager® has been fully integrated with EMIS, TPP SystmOne and Vision.
Choice of an on-screen Child Incentive animation assists in performing reliable spirometry tests by trained personnel even in preschool children.
The simple infection prevention procedure involves immersing all parts coming in the contact with patient’s exhaled air in a cold sterilising solution. The use of standard pulmonary filters is recommended.
SpiroConnect® spirometer is manufactured in the United Kingdom and comes in a soft carry pouch complete with SpiroConnect Data Manager® software on USB flash drive memory stick, Bluetooth dongle, quick start guide and the calibration certificate.

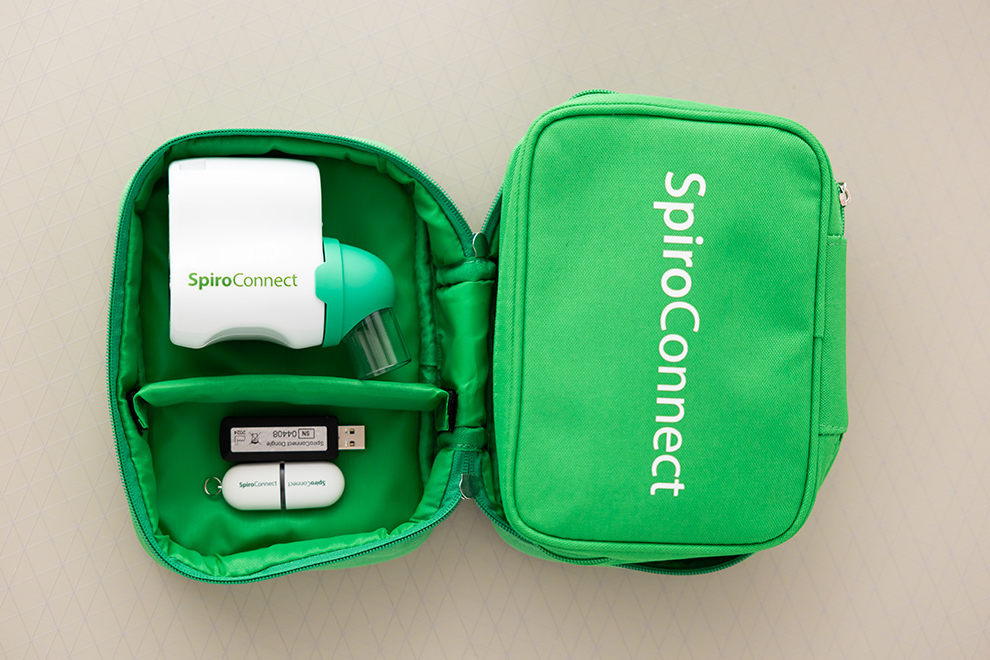
SpiroConnect®
Specifications:
| Transducer Type | Bi-directional high sensitivity vertical turbine |
| Measurements | VC, FEV0.75, FEV1, FEV3, FEV6, FVC, PEF, FEF25 (MEF75), FEF50 (MEF50), FEF75 (MEF25) FEF25-75 (MMEF), FIV1, FIVC, PIF, FIF25 (MIF75) FIF50 (MIF50), FIF75 (MIF25), MET25-75, FET, EVC, IVC, IC, VT (TV), Ti, Te, IRV, ERV, Vext, FEV0.75/VC, FEV0.75/FVC, FEV1/VC, FEV1/FVC (FER) , FEV3/VC, FEV3/FVC, FEV0.75/FEV6, FEV1/FEV6, FEF50/VC, FEF50/FVC, MMEF/FVC (FEF25-75/FVC) , FIV1/FIVC (FIR), R50 (FEF50/FIF50), Ti/Ttot, VT/Ti (TV/Ti), Lung Age, MVVind (FEV1x0.35) |
| Accuracy | To ISO 26782 and ATS 2005 standards |
| Sensitivity | Better than 0.025l/s |
| Power Supply | 2 x AA size Alkaline primary cells |
| Operating Current | 110 mA peak |
| Battery Life | Alkaline cells, greater than 100 measurement cycles |
| Dimensions | 55mm(W) x 100mm(D) x 110mm(H) |
| Weight (including batteries) |
200g |
| Operating Conditions | 10°C to 38°C, 15% to 95% RH, non-condensing, altitude up to 3,000m |
| Transport and Storage Conditions |
-20°C to 70°C, 15% to 95% RH, non-condensing |
| Lifetime | 5 years |
| Flow Range | 0- 14l/s |
| Volume Range | 0 to 8l |
| PC Software Requirements | A Microsoft Windows based PC is required, with hardware that meets the minimum specifications below: Processor: 1 GHz or above RAM: 512 MB or more Free Disk Space: 100 MB Video: 1024 x 768 minimum resolution USB: One free USB port is required OS: Win XP, Win 7, Win 8 and Win 10 |